Editorial Contributions & Industry Insights
As an international key opinion leader with extensive experience in the healthcare ecosystem, I have been at the forefront of shaping industry discussions and policy. Since 2018, I have contributed to PharmaBoardroom, offering in-depth analysis and insights on emerging trends, regulatory changes, and strategic developments within the global healthcare landscape. My work is grounded in a deep understanding of the industry's complexities, making me a trusted voice for stakeholders across the healthcare spectrum.
Featured Posts

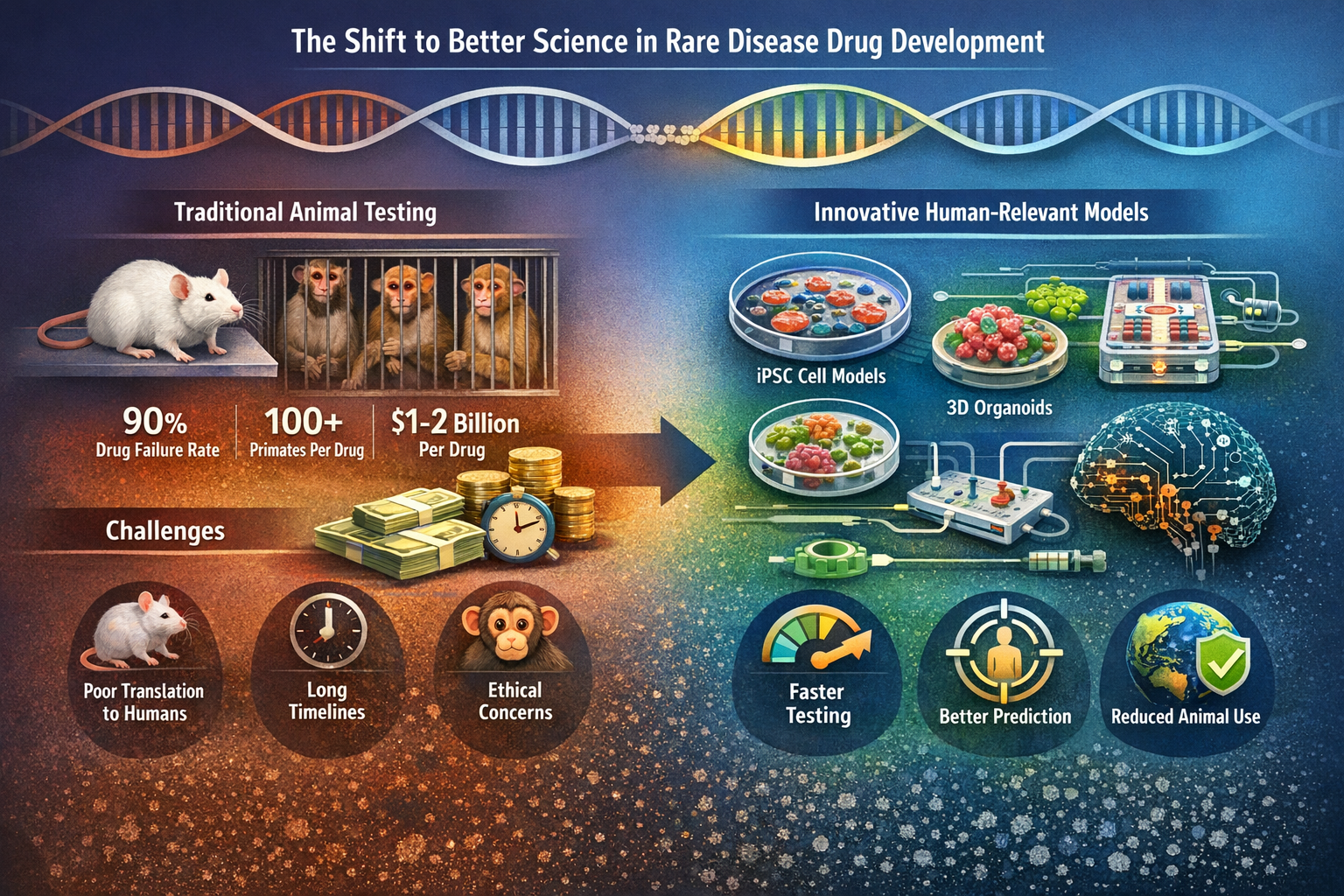
Better Science, Faster Answers: What Rare Disease Communities Need to Know About NAMs
Rare disease communities are demanding faster cures—but defending a system where over 90% of drugs fail to translate from animals to humans won’t get us there. This article breaks down why FDA is shifting toward human-relevant science, what NAMs really mean, and why better prediction—not more of the same—is the path to real progress.
A Smarter Default: Why the FDA’s New Evidence Pathway Reflects How Science Actually Works
In February 2026, the FDA announced that its longstanding requirement for two adequate and well-controlled clinical trials as the default for drug approval will be replaced with a new paradigm: one robust trial supplemented by confirmatory evidence, which may include mechanistic, observational, or real-world data. This change is intended to better reflect how modern science operates and to accelerate access to promising therapies, particularly for rare, underserved conditions.
This evolution aligns with broader updates in regulatory science. The FDA’s Advancing Real-World Evidence Program seeks to improve the quality and regulatory acceptability of real-world evidence (RWE) for new therapeutic indications and post-approval commitments, reflecting a sustained commitment to integrating data outside traditional trials into decision making.
Complementing this regulatory shift, the Promising Pathway Act 2.0—introduced in the 118th Congress—explicitly authorizes a conditional approval pathway for drugs targeting rare, serious, and progressive diseases. Under the act, sponsors may use real-world data and external sources, including natural history registries, to support applications for conditional approval, subject to requirements for subsequent evidence generation.
This legislative and regulatory flexibility is especially critical in rare disease research. Traditional clinical trial paradigms often struggle to accommodate small populations, heterogeneous phenotypes, and practical limits on enrollment and control groups. A recent systematic review in the Orphanet Journal of Rare Diseases highlights how RWE has already supported regulatory decisions for rare disease therapies, underscoring the real-world applicability of these evolving frameworks.
Indeed, scientific discovery in rare neurodevelopmental disorders frequently begins with keen observation rather than conventional endpoints. For example, patients with SYNGAP1 haploinsufficiency, a condition characterized by intellectual disability and epilepsy, often exhibit unusually high pain thresholds—a sensory characteristic now linked to circuit-specific disruption in somatosensory cortex neurons in animal models. These insights, born from patient observation and laboratory follow-up, illustrate how real-world phenomena can drive meaningful biological understanding that informs therapeutic strategies.
Together, these regulatory, legislative, and scientific developments reflect a more nuanced and patient-centered approach to evidence—one that values meaningful biological insight and real-world impact as much as traditional trial endpoints.
Subscribe to our newsletter.
Sign up with your email address to receive news and updates.



Rare disease communities are demanding faster cures—but defending a system where over 90% of drugs fail to translate from animals to humans won’t get us there. This article breaks down why FDA is shifting toward human-relevant science, what NAMs really mean, and why better prediction—not more of the same—is the path to real progress.