Editorial Contributions & Industry Insights
As an international key opinion leader with extensive experience in the healthcare ecosystem, I have been at the forefront of shaping industry discussions and policy. Since 2018, I have contributed to PharmaBoardroom, offering in-depth analysis and insights on emerging trends, regulatory changes, and strategic developments within the global healthcare landscape. My work is grounded in a deep understanding of the industry's complexities, making me a trusted voice for stakeholders across the healthcare spectrum.
Featured Posts

Better Science, Faster Answers: What Rare Disease Communities Need to Know About NAMs
Rare disease communities are demanding faster cures—but defending a system where over 90% of drugs fail to translate from animals to humans won’t get us there. This article breaks down why FDA is shifting toward human-relevant science, what NAMs really mean, and why better prediction—not more of the same—is the path to real progress.
A Smarter Default: Why the FDA’s New Evidence Pathway Reflects How Science Actually Works
In February 2026, the FDA announced that its longstanding requirement for two adequate and well-controlled clinical trials as the default for drug approval will be replaced with a new paradigm: one robust trial supplemented by confirmatory evidence, which may include mechanistic, observational, or real-world data. This change is intended to better reflect how modern science operates and to accelerate access to promising therapies, particularly for rare, underserved conditions.
This evolution aligns with broader updates in regulatory science. The FDA’s Advancing Real-World Evidence Program seeks to improve the quality and regulatory acceptability of real-world evidence (RWE) for new therapeutic indications and post-approval commitments, reflecting a sustained commitment to integrating data outside traditional trials into decision making.
Complementing this regulatory shift, the Promising Pathway Act 2.0—introduced in the 118th Congress—explicitly authorizes a conditional approval pathway for drugs targeting rare, serious, and progressive diseases. Under the act, sponsors may use real-world data and external sources, including natural history registries, to support applications for conditional approval, subject to requirements for subsequent evidence generation.
This legislative and regulatory flexibility is especially critical in rare disease research. Traditional clinical trial paradigms often struggle to accommodate small populations, heterogeneous phenotypes, and practical limits on enrollment and control groups. A recent systematic review in the Orphanet Journal of Rare Diseases highlights how RWE has already supported regulatory decisions for rare disease therapies, underscoring the real-world applicability of these evolving frameworks.
Indeed, scientific discovery in rare neurodevelopmental disorders frequently begins with keen observation rather than conventional endpoints. For example, patients with SYNGAP1 haploinsufficiency, a condition characterized by intellectual disability and epilepsy, often exhibit unusually high pain thresholds—a sensory characteristic now linked to circuit-specific disruption in somatosensory cortex neurons in animal models. These insights, born from patient observation and laboratory follow-up, illustrate how real-world phenomena can drive meaningful biological understanding that informs therapeutic strategies.
Together, these regulatory, legislative, and scientific developments reflect a more nuanced and patient-centered approach to evidence—one that values meaningful biological insight and real-world impact as much as traditional trial endpoints.
From Lab to Battlefield: How the Department of War Is Rebuilding the Technology Transfer Pipeline
Innovation has never been the Department of War’s problem. Transition has.
Despite billions invested annually in cutting-edge research, too many defense-origin technologies stall before reaching the field, the market, or the warfighter. At the Defense Patent Holiday, DOW leaders made clear that this is changing. Technology transfer is being rebuilt as a strategic capability—one that prioritizes speed, commercial viability, and scale. This article explores what’s driving that shift and what it means for organizations navigating the new defense innovation landscape.
Where Pharmaceutical Innovation Meets National Defense Strategy
Where Pharmaceutical Innovation Meets National Defense Strategy
Pharmaceutical innovation now plays a critical role in national security—far beyond emergency stockpiles or reactive countermeasures. As biological threats, supply chain vulnerabilities, and warfighter readiness rise to the forefront of defense priorities, companies seeking to engage the federal government must do more than innovate—they must align.
Drawing on deep experience across defense, health, and policy ecosystems, Monica Weldon Consulting helps pharmaceutical and biotechnology companies translate cutting-edge science into mission-relevant capabilities. By aligning innovation with presidential priorities, agency agendas, and defense acquisition pathways, we guide organizations through complex interagency landscapes with clarity and purpose.
This work isn’t about chasing contracts. It’s about strategic positioning, risk-aware engagement, and building durable partnerships that advance both national defense and scientific progress.
Why We Must Treat Fentanyl as a Chemical Threat — and What Strategic Action Looks Like
Fentanyl has rightly been framed as a public health emergency. But that framing alone is no longer sufficient.
Illicit fentanyl now sits at the intersection of public health, national security, and chemical threat preparedness. Its extreme potency, ease of synthesis, and global availability create a dual-use risk that adversaries could exploit far beyond traditional drug trafficking. Former Department of Defense officials have already warned that terrorist groups may one day attempt to aerosolize fentanyl or its analogues—an alarming prospect with real historical precedent.
In 2002, an aerosolized fentanyl-based compound was used during the Moscow theater hostage crisis, killing more than 130 civilians. Today, similar compounds are more accessible, more potent, and embedded in illicit supply chains that span the globe. The question is no longer whether this risk exists—but whether our preparedness frameworks are aligned to confront it.
Treating fentanyl as a potential chemical threat does not criminalize medicine or undermine addiction treatment. It strengthens deterrence, improves readiness, and ensures that medical countermeasures, detection technologies, and defense doctrine keep pace with evolving threats.
Preparedness is not panic. It is policy maturity.
The full article outlines what strategic action looks like—from modernizing medical countermeasure development to investing in detection and deterrence technologies and leading international norms that close dangerous legal gaps.
Fentanyl: From Public Health Crisis to National Security Threat
Fentanyl was originally synthesized in the 1960s as a medical opioid for severe pain and anesthesia, but today its illicit form dominates the U.S. opioid supply. Its extreme potency means that a lethal dose can be as little as two milligrams, making it highly dangerous. Because fentanyl is synthetically produced, there are no crop or seasonal limitations, allowing for year-round manufacture. The low cost of production means millions of doses can be created from just a few kilograms of precursor chemicals. Additionally, its easy distribution—tiny quantities can be shipped in envelopes, vehicles, or mail—makes it especially attractive to traffickers and difficult for authorities to intercept.
From Engagement to Evidence: Building a Holistic Measure of Patient-Focused Drug Development
While Patient-Focused Drug Development (PFDD) has elevated the patient voice in healthcare, significant gaps remain in translating these insights into measurable outcomes. True patient centricity goes beyond consultation—it requires embedding patient perspectives into every stage of drug development, from trial design to post-market evaluation. Yet, challenges persist: inconsistent patient engagement, lack of standardized metrics, and limited frameworks to assess the impact of patient input on decision-making. Measuring the success of patient-centric initiatives demands clear indicators, such as improvements in patient-reported outcomes, enhanced treatment adherence, and tangible changes in clinical or regulatory decisions. Only by identifying these gaps and establishing robust metrics can we ensure that patient-centered strategies deliver meaningful results rather than symbolic gestures.
CON Laws and the ACA — The Next Frontier for Affordable Healthcare Reform
More than a decade after the Affordable Care Act expanded coverage for millions, a harsh reality remains: coverage does not equal access. Premiums continue to rise, rural care deserts are widening, and consolidated insurers and middlemen dominate every layer of the system. In this article, I explore the ACA’s most overlooked weakness—its failure to confront the structural barriers that suppress competition. From Certificate-of-Need laws to PBM pricing distortions and marketplace consolidation, these forces inflate costs and restrict care long before patients reach the exam room. To achieve true affordability and access, we must finish the job the ACA started by opening the system to innovation, transparency, and competition.
A Manufacturing-Commons for America’s Medicines
COVID-19 exposed a fragile pharmaceutical supply chain — hollowed out by decades of offshoring and rising costs. Today, the U.S. faces a choice: rebuild domestic drug manufacturing or remain dependent on foreign factories. A bold, practical solution is emerging — National Centers of Excellence for Pharmaceutical Manufacturing — shared regional hubs that provide GMP-certified facilities, workforce training, and surge capacity to ensure secure, affordable medicines made on American soil. Supported by President Trump’s recent Executive Order to restore domestic production, these centers could revive our industrial base, protect national security, and make drugs more accessible. The next crisis won’t wait — and neither should we.
A Call for Collective Responsibility in Rare Disease Communities
Drawing on her experience as rare disease patient advocacy group SYNGAP1 Foundation’s founder and former president, Monica Weldon argues for the need to standardize genomic testing practices, warns against the possible ethical implications of individualized medicine, and makes a cause for a more holistic approach within rare disease communities.
Efficacy or Economics? The US Promising Pathways Act 2.0 and the Future of Innovation
The Promising Pathways Act 2.0 is a newly proposed bill that aims to expedite and streamline approval pathways for rare disease therapies in the US. The Act is, however, facing a number of challenges in terms of implementation. The sponsors of these niche therapies tend to be small and medium-sized biotech firms which are unable to compete with the financial might of Big Pharma and also struggle to deal with the myriad regulatory hurdles they need to jump over. Monica Weldon, a frequent PharmaBoardroom contributor and rare disease patient advocate, who helped craft the bill, explains more.
Subscribe to our newsletter.
Sign up with your email address to receive news and updates.



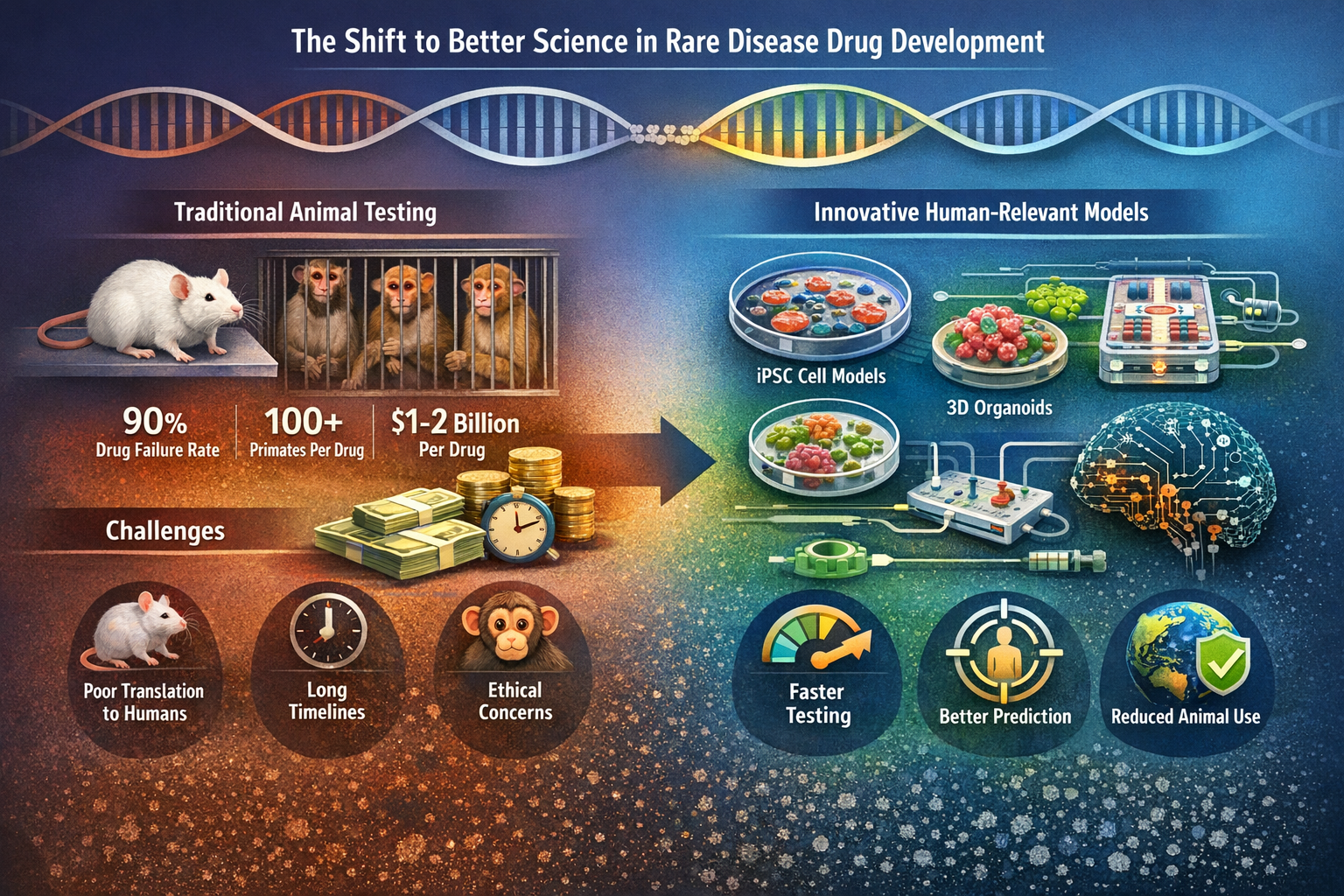






Rare disease communities are demanding faster cures—but defending a system where over 90% of drugs fail to translate from animals to humans won’t get us there. This article breaks down why FDA is shifting toward human-relevant science, what NAMs really mean, and why better prediction—not more of the same—is the path to real progress.